Code: 41411
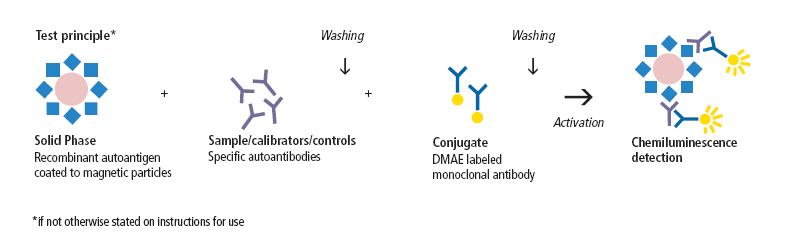
Parameter/assay principle: Quantitative detection of IgG antibodies to dsDNA, centromere B, SS-A/Ro (60 kDa and 52 kDa), SS-B/La, Sm, U1-snRNP (70 kDa, A and C), Scl-70 and Jo-1
Number of tests: 100
Sample type: Serum or plasma (EDTA, Heparin)
Sample volume: 6 µl
Incubation time: 10 + 10 minutes
Stability of calibration: 3 weeks
Cartridge stability on board: 8 weeks
Measuring range: -
Results (Index): Negative <1.0 | Positive =1.0
Clinical specificity: -
Clinical sensitivity: -
Limit of detection: -
Precision: Intra-assay CV% =4.8 | Inter-assay CV% =8.6
Relative specificity§ **: 96.0%
Relative sensitivity§ **: 96.0%
*The content of this file is for informational purposes only. The latest, updated version of the IFU, to which you should always refer, is the one accompanying the product and is to be found on the disk inside the product kit.
** versus a commercial ELISA method